Peptide Research
PATH TO PEPTIDES BEYOND OZEMPIC

PATH TO PEPTIDES BEYOND OZEMPIC
THE NEXT WAVE OF WEIGHT LOSS SCIENCE
THE HOOK
In December 2025, a clinical trial made headlines. Participants taking a new drug called retatrutide lost an average of 71 pounds — roughly 28.7% of their body weight — in just over a year. That’s nearly double what first-generation medications achieved.
But retatrutide isn’t the only new player. An oral pill called orforglipron just got fast-tracked by the FDA. And a combination drug called CagriSema is blending two different hormones into one injection. Weight loss science is moving fast. This issue breaks down what’s coming, how these drugs work differently, and what the research actually shows.
DID YOU KNOW?
Analysts estimate that weight loss medications could become a $150+ billion global market by 2031. Eli Lilly alone projects its three weight loss drugs could hit $101 billion in peak sales.
WHY THIS MATTERS
More than 40% of American adults live with obesity. That’s over 100 million people. Obesity raises the risk of heart disease, diabetes, joint problems, and dozens of other conditions.
Current medications like Ozempic and Wegovy work well for many people. But they have limits. Some patients don’t lose enough weight. Others can’t tolerate the side effects. And almost everyone faces the same barrier: weekly injections.
The next wave of drugs aims to solve these gaps — with stronger weight loss, fewer side effects, pill options, and combination approaches.
THE SCIENCE: WHAT MAKES THESE DRUGS DIFFERENT?
DRUGS DIFFERENT?
Think of your body’s hunger system like a control panel with multiple dials.
ONE DIAL: GLP-1 (CURRENT DRUGS)
Drugs like semaglutide (Ozempic/Wegovy) turn one dial: GLP-1. This hormone tells your brain you’re full, slows digestion, and helps control blood sugar. It works well. But it’s just one signal.
TWO DIALS: GLP-1 + GIP (TIRZEPATIDE)
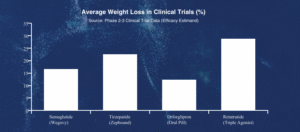
Tirzepatide (Mounjaro/Zepbound) turns two dials at once: GLP-1 and GIP. GIP is another gut hormone that helps with fat metabolism. Two signals working together produced about 22.5% average weight loss in trials.
THREE DIALS: GLP-1 + GIP + GLUCAGON (RETATRUTIDE)
Retatrutide turns three dials. It adds glucagon, a hormone that tells your liver to burn stored energy. Think of it as adding a fat-burning boost on top of appetite control. In Phase 3, it
produced 28.7% weight loss — the most ever seen in a large obesity trial.
WHAT THE RESEARCH SAYS
RETATRUTIDE: THE TRIPLE THREAT
What it is: A once-weekly injection activating three hormone receptors (GLP-1, GIP, glucagon). Made by Eli Lilly.
Key findings: The TRIUMPH-4 Phase 3 trial enrolled 445 adults. After 68 weeks, the 12 mg group lost an average of 71.2 pounds.Nearly 59% lost more than 25% of body weight. Pain scores dropped by 76%.
Side effects: Nausea, diarrhea, constipation — similar to existing drugs. A new effect called dysesthesia(unusual skin sensations) appeared in about 21% at the highest dose.
Status: Investigational. Seven more Phase 3 trials expected to report in 2026. Not yet submitted for FDA approval
ORFORGLIPRON: THE FIRST REAL WEIGHT LOSS PILL
What it is: A once-daily oral pill. Unlike oral Wegovy (which needs an empty stomach), orforglipron can be taken anytime — no food or water rules. Made by Eli Lilly.
Key findings: In ATTAIN-1, participants lost 12.4% of body weight at the highest dose. A separate trial showed it helps maintain weight loss after stopping injectable drugs.
Status: FDA application submitted late 2025. National Priority Review Voucher granted. Decision expected April 2026.
CAGRISEMA: THE COMBINATION APPROACH
What it is: A once-weekly injection combining semaglutide with cagrilintide — a long-acting form of the hormone amylin. Made by Novo Nordisk.
Key findings: In REDEFINE-2, CagriSema produced 22.7% weight loss in adults with obesity and type 2 diabetes — outperforming semaglutide alone in a population that typically loses less weight.
Status: Regulatory submission expected in 2026. Multiple Phase 3 trials ongoing.
HEAD-TO-HEAD COMPARISON
| Feature | Semaglutide (Wegovy) | Tirzepatide (Zepbound) | Orforglipron (Pill) | Retatrutide (Triple) |
|---|---|---|---|---|
| Targets | GLP-1 | GLP-1 + GIP | GLP-1 | GLP-1 + GIP + Glucagon |
| Delivery | Weekly Shot | Weekly Shot | Daily Pill | Weekly Shot |
| Avg Weight Loss | ~16.6% | ~22.5% | 12.4% | ~28.7% |
| FDA Status | Approved | Approved | Under Review | Phase 3 |
THE BIG PICTURE
We’re watching a revolution in weight loss science happen in real time. Five years ago, no medication reliably helped people lose more than 10-15%. Now drugs in trials approach 30%.
The next few years bring more options: pills, combinations, and drugs tailored to different needs. Competition between Eli Lilly and Novo Nordisk drives prices down and innovation up.
But researchers stress caution. These are powerful medications with real side effects. They work best combined with healthy eating and exercise. Long-term data is still being collected.
THE BOTTOM LINE
Weight loss science is entering a new era. Retatrutide sets new records with triple-agonist technology. Orforglipron could make daily pills as effective as injections were just a few years ago. The research is promising — but still unfolding.
WHAT TO KNOW
These drugs are not shortcuts. Research shows the best outcomes come from combining medication with lifestyle changes — better nutrition, regular exercise, and working with a healthcare provider.
Not all are available yet. Semaglutide and tirzepatide are FDA-approved. Orforglipron is under review. Retatrutide is in Phase 3 trials. CagriSema hasn’t been submitted.
Talk to your doctor. Every person responds differently. Your healthcare provider can help determine which approach makes sense for your situation.
REFERENCES
1. CDC. Adult Obesity Facts. Centers for Disease Control and Prevention, 2024.
2. Eli Lilly. TRIUMPH-4 Phase 3 Trial Results: Retatrutide. Press Release, Dec 11, 2025.
3. Giblin K, et al. Retatrutide TRIUMPH program design. Diabetes Obes Metab. 2025.
4. Jastreboff AM, et al. Retatrutide Phase 2 Results. N Engl J Med. 2023;389(6):514-526.
5. Eli Lilly. ATTAIN-1 Phase 3 Results: Orforglipron. Press Release, Aug 26, 2025.
6. Eli Lilly. ATTAIN-MAINTAIN Results: Orforglipron. Press Release, Dec 18, 2025.
7. Novo Nordisk. REDEFINE-2 CagriSema Phase 3 Results. 2025.
8. Frias JP, et al. Tirzepatide vs Semaglutide (SURMOUNT-5). N Engl J Med. 2025.
9. Wilding JPH, et al. Semaglutide Weight Management. N Engl J Med. 2021;384:989-1002.
10. FDA. Oral Wegovy (semaglutide) Approval. December 2025.
11. BioSpace. Retatrutide Phase 3 Analysis. December 12, 2025.
12. Pharmaceutical Journal. Retatrutide Weight Loss Data. December 15, 2025.
13. MedCentral. Orforglipron FDA Decision Analysis. February 2026.
14. GlobalData. Retatrutide Sales Forecast: $15.6B by 2031.
15. Truist Securities. Lilly Weight Loss Trio Peak Sales Estimate: $101B.
16. World Obesity Federation. Global Obesity Statistics. 2024.
FOR RESEARCH AND EDUCATIONAL PURPOSES ONLY
This document is intended solely for educational purposes to increase awareness of emerging scientific research. It does not constitute medical advice and should not be used to make healthcare decisions.
Regulatory Status: Semaglutide (Wegovy) and tirzepatide (Zepbound) are FDA-approved for chronic weight management. Orforglipron is under FDAreview. Retatrutide and CagriSema are investigational and not FDA-approved.
All healthcare decisions should be made in consultation with qualified medical professionals.